Meiosis is a unique type of cell division that ensures the
|
|
|
- Tracey Kelley
- 6 years ago
- Views:
Transcription
1 Transmembrane protein Sun2 is involved in tethering mammalian meiotic telomeres to the nuclear envelope Johannes Schmitt*, Ricardo Benavente*, Didier Hodzic, Christer Höög, Colin L. Stewart, and Manfred Alsheimer* *Department of Cell and Developmental Biology, Biocenter of the University of Würzburg, D Würzburg, Germany; Department of Cell Biology and Physiology, Washington University School of Medicine, St. Louis, MO 63110; Department of Cell and Molecular Biology, Karolinska Institute, SE Stockholm, Sweden; and Cancer and Developmental Biology Laboratory, National Cancer Institute, Frederick, MD Edited by Nancy Kleckner, Harvard University, Cambridge, MA, and approved March 16, 2007 (received for review October 18, 2006) Dynamic repositioning of telomeres is a unique feature of meiotic prophase I that is highly conserved among eukaryotes. At least in fission yeast it was shown to be required for proper alignment and recombination of homologous chromosomes. On entry into meiosis telomeres attach to the nuclear envelope and transiently cluster at a limited area to form a chromosomal bouquet. Telomere clustering is thought to promote chromosome recognition and stable pairing of the homologs. However, the molecular basis of telomere attachment and movement is largely unknown. Here we report that mammalian SUN-domain protein Sun2 specifically localizes to the nuclear envelope attachment sites of meiotic telomeres. Sun2 telomere association is maintained throughout the dynamic movement of telomeres. This association does not require the assembly of chromosomal axial elements or the presence of A-type lamins. Detailed EM analysis revealed that Sun2 is part of a membrane-spanning fibrillar complex that interconnects attached telomeres with cytoplasmic structures. Together with recent findings in fission yeast, our study indicates that the molecular mechanisms required for tethering meiotic telomeres and their dynamic movements during bouquet formation are conserved among eukaryotes. meiosis SUN-domain proteins telomere attachment Meiosis is a unique type of cell division that ensures the proper segregation of genetic material and the generation of viable haploid gametes. This is accomplished by a complex series of chromosomal interactions during first meiotic division that include alignment, pairing, synapsis, and recombination of the homologous chromosomes during prophase I, which in turn are prerequisites for the reductional segregation. Despite its incontestable significance for sexual reproduction and the generation of genetic variability, many questions regarding the most crucial events remain to be answered, among which is the understanding of the mechanisms that lead to specific alignment and stable pairing of the homologous chromosomes. Induction of meiosis causes a unique redistribution of telomeres, which is highly conserved among eukaryotes. During early meiotic prophase telomeres migrate to the nuclear periphery, where they become physically attached to the nuclear envelope (NE). Once attached, they start to move along the inner NE and transiently cluster at a limited area to form a so-called chromosomal bouquet (for review see refs. 1 and 2). The bouquet configuration disappears at pachytene stage, and telomeres, while still remaining attached, redisperse around the inner surface of the NE. Because of the close temporal correlation between telomere clustering and homologue pairing as well as synapsis, it was suggested that the unique telomere dynamics during meiotic prophase are required for facilitating chromosome recognition and stable pairing of the homologs (1, 3 5). This hypothesis has been supported by previous studies in yeast. In mutants that are characterized by the absence of telomere clustering, homologue pairing and recombination are significantly delayed or disrupted (6 9). Formation of a bouquet is certainly not sufficient to ensure pairing and synapsis as evidenced by several mutants that are defective for early recombination events such as formation of double-strand breaks (for review see ref. 10). Nevertheless, the bouquet mutants demonstrated that telomere clustering is essential for proper meiotic progression, most likely by supporting recognition and alignment of the homologs. However, the molecular mechanisms that are required for attachment and movement of chromosomal ends are largely unknown. In the past, several studies provided evidence that the NE for its part may play an important role in meiotic chromosome dynamics (for review see refs. 2, 11, and 12). Support for this came from observations that the NE of a meiotic cell differs remarkably from that of a somatic one. For instance, compared with somatic cells, in mammalian spermatocytes the nuclear lamina, i.e., the structural element of the NE that is critically involved in many fundamental cellular and developmental processes (for a recent review see ref. 13), shows significant differences regarding its composition and organization. Somatic lamins A, C, and B2 are absent. Instead, these cells express somatic lamin B1 together with lamin C2, a N-terminally shortened meiosis-specific variant of the lamin A gene (14 16). Contrary to somatic lamins, lamin C2 forms discontinuous domains at the NE of meiotic cells. Moreover, it is highly enriched at the attachment sites of the telomeres, indicating that in the immediate vicinity of these sites the NE becomes locally modulated. It was suggested that lamin C2 might lead to a local flexibility of the NE at the attachment sites that in turn enables the movement of telomeres and therefore supports pairing, recombination, and synapsis (17, 18). Beside the changes in its composition, at the attachment sites of meiotic chromosomes the NE exhibits some additional characteristic features as revealed by ultrastructural analyses (12, 19 21). The attachment of meiotic telomeres to the NE involves a terminal morphological specialization of the axial elements (AEs) of the synaptonemal complexes (SCs) called attachment plates. These structures appear as disk-shaped electrondense plates that are intimately associated with the nuclear face of the inner nuclear membrane (INM) (21). Adjacent to the plates the nuclear membranes are more dense than in other regions. Thin fibrils arise from the plates, some of which traverse the perinuclear space and emanate into the cytoplasm, suggesting that chromosomal ends are directly interconnected with cytoplasmic structures. Such filaments bridging the nuclear membranes have been observed at zygotene and pachytene telomeres in a variety of different Author contributions: R.B. and M.A. designed research; J.S. and M.A. performed research; D.H., C.H., and C.L.S. contributed new reagents/analytic tools; J.S., R.B., and M.A. analyzed data; and M.A. wrote the paper. The authors declare no conflict of interest. This article is a PNAS Direct Submission. Abbreviations: SPB, spindle pole body; NE, nuclear envelope; SC, synaptonemal complex; AE, axial element; LE, lateral element; INM, inner nuclear membrane. To whom correspondence should be addressed. alsheimer@biozentrum. uni-wuerzburg.de by The National Academy of Sciences of the USA PNAS May 1, 2007 vol. 104 no cgi doi pnas
2 species, but up to the present their composition and function have remained elusive (12, 19, 20). Recently, a new family of INM proteins has been identified that share a conserved SUN (Sad1/UNC-84 homology) domain. In somatic cells SUN-domain proteins were shown to be part of a protein complex spanning the nuclear membranes that provides a structural linkage between the cytoskeleton and nuclear components, which in turn is crucial for nuclear anchoring, migration, and positioning (22 27). In mammals two INM-localized SUN-domain proteins were identified, Sun1 and Sun2, that are derived from two different genes (28, 29). Both proteins contain a N-terminal nucleoplasmic domain that was found to bind nuclear lamins. The C terminus containing the conserved SUN domain extends into the perinuclear space, where it interacts with the KASH domain of both nesprin1 and nesprin2, which are outer nuclear membrane proteins that for their part are directly connected to the actin cytoskeleton (22 24). Thus, in somatic cells SUN-domain proteins appear to be at the very center of a nucleocytoplasmic bridge that is essential for nuclear motility. Since during meiotic prophase I a filamentous nucleocytoplasmic connection is established specifically at the attachment sites of meiotic telomeres, SUN-domain proteins may take part in forming these bridging filaments and therefore represent quite good candidates for being involved in tethering meiotic telomeres to the NE. In the present study we focused our interest on Sun2, and we report on its specific localization pattern and behavior in the context of meiotic chromosome dynamics. Our findings provide novel insights into meiotic chromosome dynamics, and we show further support for the existence of a nuclear cytoplasmic linkage that is suggested to be required for homologue pairing and synapsis occurring in the nuclear interior. Results During Meiotic Prophase I Sun2 Is Selectively Located at the Attachment Sites of Meiotic Chromosomes. As described above, Sun2 is part of a membrane-spanning protein complex that was suggested to interconnect the nucleoskeleton with cytoplasmic actin filaments in somatic cells. Remarkably, in meiotic cells thin fibrils have been observed at the attachment sites of telomeres that arise from the attachment plates, traverse the perinuclear space, and apparently emanate into the cytoplasm (see below). Therefore, we hypothesized that Sun2 may be involved in forming these interconnecting filaments. To test this hypothesis we investigated the localization of Sun2 in the context of meiotic telomere anchoring to the NE. Thus, we made use of an affinity-purified anti-sun2 antibody (29), which we applied to detailed immunofluorescence microscopy. Consistently with previous findings, in somatic cells of rat testes Sun2 showed a homogeneous rim-like pattern within the NE (29) (see Fig. 1A). However, in contrast to the situation in somatic interphase cells, this pattern is not maintained when cells enter meiosis. In rat as well as mouse pachytene spermatocytes we found a remarkable redistribution of Sun2. Instead of the homogeneous distribution throughout the NE, we observed a distinct dotted staining pattern with the signals nevertheless being located along the nuclear periphery (Figs. 1 B D and 2A). As shown in Fig. 1B and in agreement with our previous findings (17), such a redistribution is not seen in the case of LAP2 isoforms, which represent another group of integral proteins of the INM (for review see ref. 30). In meiotic cells that are characterized by a punctured distribution of Sun2, integral LAP2 are evenly spread throughout the NE. This indicates that the punctured distribution is a specific property of Sun2 and not a general behavior of INM proteins during meiotic prophase I (Fig. 1 B B ). Due to the fact that the punctate pattern observed with the Sun2 antibody is reminiscent of the telomere distribution pattern during meiotic prophase I (see, e.g., ref. 31), we asked whether there is a spatial relationship between Sun2 signals and the attachment sites of meiotic telomeres. In double immunofluorescence analyses using Fig. 1. Sun2 localizes to the NE attachment sites of meiotic telomeres. (A) Sun2 (green) shows a homogeneous rim-like pattern within the NE of rat somatic testicular cells. (B G) Localization of Sun2 in prophase I spermatocytes. Sun2 displays a punctate pattern in pachytene (B D) as well as leptotene (E), zygotene (F), and diplotene (G) stages. (A G ) The same cells were costained for LAP2 or SYCP3 (red). Overlays are seen in A G. All sections were labeled with the DNA-specific fluorochrome Hoechst (A G ). Some of the Sun2 spots are denoted by arrows; arrowheads indicate the midplane of nuclei. Images were taken from testis cryosections by confocal laser scanning microscopy, with the exception of D, which represents a whole-cell preparation recorded with wide-field epifluorescence microscopy. (Scale bars: 10 m.) Sun2 antibody together with an anti-sycp3 antiserum, which labels the AEs/lateral elements (LEs) of the SCs (16), we observed that Sun2 signals were closely associated with the sites where AEs/LEs make intimate contact to the NE (Fig. 1 C and D). Evaluation of 600 Sun2 signals in whole-cell preparations of rat pachytene spermatocytes recorded with wide-field epifluorescence microscopy displayed a mean of spots per nucleus ( 0.26 SD), whereby all spots were associated with ends of the bivalents (Fig. 1 D D ). This clearly indicates that virtually every SC end is decorated with a Sun2 signal (in the rat there are 40 ends corresponding to the autosomal bivalents and three ends corresponding to the partially paired X and Y chromosomes). By closer inspection using CELL BIOLOGY Schmitt et al. PNAS May 1, 2007 vol. 104 no
3 Fig. 2. Sun2 localization is independent of A-type lamins and does not require AE/LE assembly. Shown is immunolocalization of Sun2 (green) in spermatocytes (A, B, and D) or somatic testicular cells (C) from wild-type (A), Lmna / (B and C), and Sycp3 / (D) mice. Sections were costained with antibodies against SYCP3 (A C) or alternatively with CREST serum, which labels centromeres that, in the mouse, are located at one end of each chromosome (D) (red). All sections were labeled with the DNA-specific fluorochrome Hoechst (A F ). Overlays are seen in A D. (Scale bars: 10 m.) confocal microscopy we could find Sun2 residing directly on top of the SC ends (Inset in Fig. 1C ), pointing to a close contact between Sun2 and the chromosomal ends at the NE. This observation could be corroborated by immunogold electron microscopy. In mouse pachytene spermatocytes we found strong Sun2 labeling within NE domains that are associated with the attachment sites of telomeres, whereas adjacent NE areas as well as the surrounding cytoplasm and nucleoplasm were virtually free of gold particles (Fig. 3 B D). Remarkably, most gold particles decorated thin filaments that arise form the attachment plates (Fig. 3 B D), suggesting that Sun2 is part of the filamentous structures that originate from the attachment plates, traverse the perinuclear space, and protrude into the cytoplasm (Fig. 3A) (19, 20). Sun2 Redistribution Within the NE Is Temporally Associated With Meiotic Telomere Dynamics. One of the most striking observations during early meiotic prophase is that telomeres, once attached, start to move on the plane of the NE and congregate at one pole to form a so-called bouquet (1, 2). The observations described above clearly demonstrate that in pachytene stage of meiotic prophase I Sun2 localizes to the NE attachment sites of meiotic chromosomes. Given the role that Sun2 connects nuclear components to the actin cytoskeleton via its interaction with nesprin2 Giant (nesp2g) (22, 23), and because of the fact that SUN-domain proteins are critically involved in nuclear motions (25, 32), we propose that Sun2 might also be crucially involved in telomere movement during bouquet formation. If so, Sun2 localization has to be closely related to the attachment sites of the chromosomal ends during the whole process when telomeres move actively along the plane of the NE. To address this issue we started to investigate the specific behavior of Sun2 in the context of meiotic telomere dynamics. Remarkably, as early as leptotene stage when AEs become formed and telomeres, having attached to the NE, actively congregate at one pole (for review see Fig. 3. Immunogold EM examination of Sun2 in mouse spermatocytes. (A) Electron microscopic analysis of a telomere attachment site in pachytene. Ch, chromatin; CE, central element of the SC. (B D) Electron microscopic immunolocalization of Sun2. Attachment plates are denoted by arrowheads; arrows indicate some of the cytoplasmic fibrils that are attached to the NE. (Scale bar: 100 nm.) ref. 1), Sun2 already displayed a discontinuous, rather punctate pattern along the nuclear periphery (Fig. 1E). This pattern reflects the above-described situation in pachytene cells in principle (Figs. 1 B D and 2A). However, in contrast to the pattern in pachytene and concomitant with the specific congregation of telomeres during early meiotic prophase, Sun2-containing domains were not found evenly distributed throughout the NE, but they were clearly polarized to one half of the nucleus (Fig. 1E). A similar behavior of Sun2 could be observed in early zygotene stage as well, i.e., Sun2- containing spots were restricted to one side of the cell nucleus (Fig. 1F). Moreover, during the latter stage Sun2 could be found exclusively residing at the sites where the now-established AEs are in contact with the NE (Fig. 1F). Finally, after resolution of the bouquet configuration, from pachytene to diplotene stage Sun2 domains, although being directly associated with the chromosomal ends, are randomly distributed throughout the NE (Figs. 1 B D and G, 2A, and 3). Taken together, these results provide strong evidence that the close contact between Sun2-containing structures and the attachment sites is established early in meiotic prophase and maintained throughout the process of dynamic movement of meiotic telomeres. Sun2 Localization in Spermatocytes Is Independent of A-Type Lamins and Does Not Require AE/LE Assembly. In a previous study we demonstrated that meiotic cells express a single A-type lamin cgi doi pnas Schmitt et al.
4 isoform, namely the meiosis-specific lamin C2, which shows a remarkable behavior as it is discontinuously distributed within the NE, highly enriched at the attachment sites of meiotic chromosomes (16, 17). Because it was shown that somatic A-type lamins interact with Sun2 (23), Sun2 localization to the attachment plates might be a direct consequence of lamin C2 enrichment at these sites. To test this possibility we investigated Sun2 distribution in mice lacking A-type lamins (33). Previously it was shown that in mammalian somatic cells the absence of A-type lamins has only weak consequence for Sun2 distribution, if any (see refs and 34). Consistently, we found that Sun2 distribution in somatic cells of Lmna / testes is not affected, i.e., Sun2 showed a continuous rim-like pattern throughout the NE (Fig. 2C). Similar to the situation in somatic cells, within meiotic prophase cells the absence of the meiotic A-type lamin, lamin C2, has virtually no effect on Sun2 localization as well. We could find that Sun2 was still distributed in a punctured pattern whereby the close association with the attachment sites of meiotic chromosomes was maintained (Fig. 2B). Thus, our results clearly demonstrate that meiosis-specific redistribution of Sun2 and its association with chromosomal ends are independent of lamin C2 enrichment at these sites. Due to the fact that AEs/LEs were shown to be in continuity with the filaments emanating into the cytoplasm (for review see ref. 1), we asked whether the formation of AEs might be responsible for the recruitment of Sun2 to the attachment sites of meiotic chromosomes. Therefore, we analyzed Sun2 distribution in spermatocytes of mice lacking SYCP3, a protein that represents the major structural component of the AEs/LEs (21, 35). In Sycp3 / males cells enter meiosis, but meiotic progression is impaired, leading to apoptotic cell death in zygotene stage of meiotic prophase I (35). Furthermore, ultrastructural analysis of Sycp3 / spermatocytes revealed that SC formation is disrupted with AE/LEs being absent (21). In analyzing Sun2 localization in male Sycp3 / meiocytes, we could find that Sun2 distribution is indistinguishable between normal spermatocytes and Sycp3 / spermatocytes lacking AE/ LEs. Similar to the wild-type situation, Sun2 is concentrated in small-sized spots residing at the attachment sites of telomeres (Fig. 2 A and D). Thus, this result provides strong evidence that Sun2 does not require AE assembly for its localization to the attachment sites of chromosomal ends, which is consistent with our previous observation that the fibrillar material extending into the cytoplasm can still be found associated with the attachment plates in Sycp3 / spermatocytes (21). Discussion In previous studies in yeast it has been demonstrated that meiotic telomere clustering is mandatory for meiotic progression (6 9). However, up to the present the molecular requirements for NE attachment and congregation of chromosomal ends during meiotic prophase I remained largely unknown. Progress in this issue came from recent observations in fission yeast. In meiosis of Schizosaccharomyces pombe, telomeres become clustered at the NE adjacent to the spindle pole body (SPB) (36). This clustering is mediated by the SPB-associated protein Sad1p, which is thought to function as a transmembrane linker involved in interconnecting meiotic telomeres with the SPB (37, 38). Sad1p belongs to a novel group of INM proteins, termed SUN-domain proteins, that are found across all eukaryotes. SUN-domain proteins were shown to be required for nuclear anchoring, migration, and positioning in somatic cells. Thus, they hold a key position at the nuclear cytoplasmic interface, where they appear to be part of a transmembrane complex that links nuclear components to the cytoskeleton (for recent overview see refs. 27 and 39). Recently, Tomita and Cooper (40) hypothesized that, like Sad1p in S. pombe, SUN-domain proteins may be essentially involved in mammalian bouquet formation as well, i.e., in NE tethering and directed movement of telomeres. However, until now the expression and behavior of SUN-domain proteins in mammalian meiotic cells remained speculative. In our present study we could demonstrate that with Sun2 at least one of the mammalian SUN-domain proteins is expressed during mammalian male meiosis. Sun2 was previously identified as an INM protein that in somatic cells is homogeneously distributed throughout the NE (29). However, within meiotic cells it shows a remarkably different behavior, as we could find Sun2 exclusively residing at the attachment sites of telomeres. This specific Sun2 telomere association appears as early as leptotene stage and is maintained throughout the dynamic movement of chromosomal ends. Based on these results we suggest that within meiotic cells Sun2 represents a constitutive component of the meiotic attachment complex that structurally links telomeres to the NE. Consistently, by EM immunolocalization we found that Sun2 is concentrated at membranespanning fibrillar complexes that connect to telomeres along the inner nuclear surface, traverse the perinuclear space, and radiate into the cytoplasm. Such telomere-associated filaments have been observed in a variety of different species, but up to the present their function and composition have remained elusive (12, 19, 20). Hence, our results now provide a hint for the molecular nature of these filaments, with Sun2 appearing to be a central component. Moreover, our findings suggest that the mammalian meiotic cell recruits NE components also expressed in somatic cells for tethering telomeres to the NE, and they provide clear support for the model described above whereby in mammals, with Sun2, a SUNdomain protein appears to be implicated in forming a meiotic telomere attachment complex that in turn might be functional in telomere clustering (ref. 40 and see below). Recently, Chikashige et al. (38) presented compelling evidence that in fission yeast SUN-domain protein Sad1p holds a key position in meiotic telomere clustering. During meiotic prophase, the nucleoplasmic domain of Sad1p interacts with Bqt1p, which in turn binds telomere-associated Rap1p in the presence of Bqt2p, resulting in a structural linkage between telomeres and the NE (38, 41). On the other hand, the C-terminal conserved SUN domain of Sad1p, which most likely is located within the perinuclear space, was shown to interact with the KASH domain of Kms1p, an outer nuclear membrane protein that directly connects to the SPB (32, 42 44). Thus, Sad1p appears to hold a central position in a linker complex that interconnects meiotic telomeres with cytoskeletal structures. Our findings in the present study suggest a similar function for Sun2 in mammalian meiotic cells: (i) during meiotic prophase I, Sun2 is strictly associated with telomeres, which resembles Sad1p behavior in S. pombe; (ii) during bouquet formation telomeres that are linked to the Sun2-containing attachment complex congregate near the centrosome, the animal equivalent of the yeast SPB (2, 21); and (iii) Sun2 takes part in forming fibrillar structures that provide a nucleocytoplasmic bridge, hence interconnecting telomeres with cytoplasmic structures. Interestingly, it was recently found that Sun2 interacts with the KASH domain of outer nuclear membrane protein nesp2g to form a bridge across the NE (23), thus reflecting the Sad1p-Kms1p NE bridging complex formed in fission yeast. Taken together, the overt correlation between S. pombe Sad1p and mammalian Sun2 suggests that telomere anchoring and clustering follow a general mechanism that is conserved upon eukaryotes. In agreement with this assumption, in Caenorhabditis elegans SUN-1/matefin, a germ line-specific SUNdomain protein, was found to be essential for germ cell viability; however, up to the present it is completely unknown whether SUN-1/matefin is functionally involved in telomere attachment and movement (45). In mammals the INM of somatic cells contains a second SUN-domain protein, namely Sun1, which was suggested to have overlapping functions with Sun2 (23, 28). Up to now, it is not known whether Sun1 is expressed in the germ line as well. Thus, this point has to be clarified before a putative involvement of Sun1 in meiotic events can be postulated. CELL BIOLOGY Schmitt et al. PNAS May 1, 2007 vol. 104 no
5 Fig. 4. Model for the organization of the NE at the attachment sites of meiotic chromosomes. In meiotic cells Sun2 is exclusively located at the attachment plates, where it is involved in tethering meiotic telomeres to the NE. Within the perinuclear space its C-terminal SUN domain binds to the KASH domain of nesp2g, thus forming a stable fibrillar complex bridging both nuclear membranes. Because nesp2g interacts with cytoplasmic actin via its actin binding domain, the formation of such a complex would link telomeres to the actin cytoskeleton. This model is consistent with recent findings that identified an actin-dependent mechanism for bouquet formation. AP, attachment plate; CE, central element of the SC; NPC, nuclear pore complex; PNS, perinuclear space; ONM, outer nuclear membrane. Because Sun2 was shown to interact with nesp2g and given that nesp2g connects to the actin cytoskeleton (23, 46) we propose a model describing NE attachment and movement of meiotic telomeres in mammals (Fig. 4). Upon entry into meiosis telomeres move toward the nuclear periphery to contact the NE. Then, Sun2 initially dispersed in the INM becomes recruited to telomeres by a yet-unknown mechanism, forming a mechanically stable telomere INM connection. As we have shown here, this specific Sun2 redistribution is independent of AE formation as well as of lamin C2 enrichment at the attachment sites and thus requires other proteins, perhaps yet unidentified orthologues of S. pombe Bqt1p and Bqt2p that were shown to recruit Sad1p, hence tethering telomeres to the NE (38, 40, 41). Finally, through its interaction with nesp2g, Sun2 could link telomeres directly to the actin cytoskeleton. Thus, our model would favor a cytoplasmic actin-based mechanism that generates the forces needed for meiotic telomere dynamics, including their congregation during bouquet formation. Notably, in budding yeast inhibition of actin polymerization leads to disruption of telomere clustering, indicating the existence of such a mechanism; however, it is not known whether cytoplasmic or nuclear actin is involved in these processes (47). Also unknown is whether actin-dependent bouquet formation applies to other species like mammals. Beside a putative role in telomere clustering, linkage of telomeres to the cytoskeleton via the Sun2-containing attachment complex might contribute to more basic nuclear events occurring during early meiotic prophase I, such as the vigorous nuclear rotations and chromosomal oscillations observed during leptotene/zygotene in the rat that are thought to be required for alignment of the homologs (48). The movements of the entire nucleus could be induced by simply pulling at individual and/or groups of telomeres with respect to the centrosome, hence placing the affected telomeres near the centrosome (see also ref. 31). However, further analyses are required to clarify these important questions. Materials and Methods Animals and Tissue Preparation. Male Wistar rats (39 42 days old) were obtained from Charles River (Sulzfeld, Germany). Wild-type, Lmna /, and Sycp3 / C57BL/6 mice used in this study were described previously (33, 35). Animals were anesthetized and killed with CO 2. Testes were immediately resected, shock-frozen for 5 min in 2-methylbutane (Carl Roth, Karlsruhe, Germany), cooled with liquid nitrogen to 140 C, and stored at 70 C in the presence of 2-methylbutane until further use. For immunofluorescence microscopy, 6- m-thin cryosections were made with a Reichert-Jung Kryostat 2800 Frigocut-E (Leica, Bensheim, Germany). For testis suspensions, fresh rat testes were transferred in a glass Petri dish containing ice-cold PBS (140 mm NaCl/2.6 mm KCl/6.4 mm Na 2 HPO 4 /1.4 mm KH 2 PO 4, ph 7.4). After removing the tunica albuginea, seminiferous tubules were cut into small pieces cgi doi pnas Schmitt et al.
6 and carefully resuspended. To obtain a fraction of singularized cells enriched with spermatogenic stages, the suspension was filtered through a nylon filter (mesh size 30 m). The cells were washed once with ice-cold PBS, resuspended in a suitable volume of PBS, and transferred to Superfrost Plus coverslips (Menzel, Braunschweig, Germany). Attached cells were then fixed as described below. Antibodies. The following primary antibodies were used: affinitypurified rabbit anti-sun2 antiserum (29), affinity-purified guinea pig anti-sycp3 (16), mab 13d4 against LAP2 (49), and human CREST serum (gift from Georg Krohne University of Würzburg, Germany). Secondary antibodies conjugated to Cy2 or Texas red were purchased from Dianova (Hamburg, Germany). Immunofluorescence Microscopy. Cryosections or cells from testes suspensions were fixed for 5 min in PBS containing 1% formaldehyde and permeabilized for 10 min in PBS containing 0.1% Triton X-100. After blocking for 1 h with PBT(PBS containing 1.5% BSA and 0.1% Tween 20), slides were incubated simultaneously with selected primary antibodies for 20 min. Then specimens were washed twice in PBS, and bound antibodies were detected with corresponding secondary antibodies. DNA was visualized with the fluorochrome Hoechst (Hoechst, Frankfurt, Germany). After a final washing in PBS, the cells were embedded in Mowiol. Epifluorescence analyses were done with an Axiophot stereo fluorescence microscope (Zeiss, Jena, Germany) equipped with a Pixelfly digital system (PCO Computer Optics, Kelheim, Germany). For confocal microscopy, we used a Leica TCS-SP2 AOBS confocal laser scanning microscope. Settings were identical for all scanning procedures. Single optical sections were taken with a HCX PL APO lbd.bl 63/1.4 oil-immersion objective with zoom 4, a pinhole setting at m, and 4 accumulations. All fluorochromes were scanned sequentially. Cy2 was excited with a wavelength of 488 nm, Texas red with 561 nm, and Hoechst with 405 nm. Series of three sections with a step size of m were taken. The maximum projection algorithm of the Leica software was then used to calculate information sectored in different ranks to a two-dimensional projection with a defined thickness of m. The obtained data were processed by using Photoshop (Adobe Systems, San Jose, CA). EM. Mouse testes were fixed in 2.5% cacodylate buffered glutaraldehyde (1 h, 4 C) and then postfixed with 1% osmium tetroxide (1 h). After overnight staining with 0.5% uranyl acetate, testes were dehydrated in ethanol series and embedded in Epon. Ultrathin sections of Epon-embedded testes were double-stained with uranyl acetate and lead citrate according to standard procedures. For immunogold localization of Sun2, mouse testis cryosections were processed similar to the immunofluorescence protocol with the exception that Fluoro Nanogold anti-rabbit (Nanoprobes, New York, NY) was used as secondary antibody. After washing in PBS, the cryosections were refixed for 10 min in 2% glutaraldehyde in PBS and washed several times in distilled water. For silver enhancement, the Aurion R-GENT SE-EM Kit (Aurion, Wageningen, The Netherlands) was used. Sections were dehydrated in ethanol series and embedded in Epon according to standard protocols. Ultrathin sections were stained as described above. Micrographs were obtained with a Zeiss EM-10 electron microscope. We thank Elisabeth Meyer-Natus for excellent technical assistance. This work was supported by Deutsche Forschungsgemeinschaft Grants Al 1090/ 1 1 (to M.A.) and Be 1168/6 2 (to R.B.) and Graduate School 1048 of the University of Würzburg Grant A4 (to R.B.). D.H. was supported by a grant from the Muscular Dystrophy Association, and C.H. was supported by the Swedish Research Council. 1. Zickler D, Kleckner N (1998) Annu Rev Genet 32: Scherthan H (2001) Nat Rev Mol Cell Biol 2: Loidl J (1990) Genome 33: Dernburg AF, Sedat JW, Cande WZ, Bass HW (1995) in Telomeres, eds Blackburn EH, Greider CW (Cold Spring Harbor Lab Press, Cold Spring Harbor, NY), pp Niwa O, Shimanuki M, Miki F (2000) EMBO J 19: Cooper JP, Watanabe Y, Nurse P (1998) Nature 392: Nimmo ER, Pidoux AL, Perry PE, Allshire RC (1998) Nature 392: Trelles-Sticken E, Dresser ME, Scherthan H (2000) J Cell Biol 151: Ding DQ, Yamamoto A, Haraguchi T, Hiraoka Y (2004) Dev Cell 6: Lichten M (2001) Curr Biol 11:R253 R Wilson EB (1925) The Cell in Development and Heredity (Macmillan, New York). 12. Holm PB (1977) Carlsberg Res Commun 42: Gruenbaum Y, Margalit A, Goldman RD, Shumaker DK, Wilson KL (2005) Nat Rev Mol Cell Biol 6: Smith A, Benavente R (1992) Differentiation 52: Furukawa K, Inagaki H, Hotta Y (1994) Exp Cell Res 212: Alsheimer M, Benavente R (1996) Exp Cell Res 228: Alsheimer M, von Glasenapp E, Hock R, Benavente R (1999) Mol Biol Cell 10: Alsheimer M, Liebe B, Sewell L, Stewart CL, Scherthan H, Benavente R (2004) J Cell Sci 117: Esponda P, Gimenez-Martin G (1972) Chromosoma 38: Benavente R, Alsheimer M, von Glasenapp E (2004) Chromosomes Today 14: Liebe B, Alsheimer M, Höög C, Benavente R, Scherthan H (2004) Mol Biol Cell 15: Padmakumar VC, Libotte T, Lu W, Zaim H, Abraham S, Noegel AA, Gotzmann J, Foisner R, Karakesisoglou I (2005) J Cell Sci 118: Crisp M, Liu Q, Roux K, Rattner JB, Shanahan C, Burke B, Stahl PD, Hodzic D (2006) J Cell Biol 172: Haque F, Lloyd DJ, Smallwood DT, Dent CL, Shanahan CM, Fry AM, Trembath RC, Shackleton S (2006) Mol Cell Biol 26: Lee KK, Starr D, Cohen M, Liu J, Han M, Wilson KL, Gruenbaum Y (2002) Mol Biol Cell 13: Starr DA, Han M (2003) J Cell Sci 116: Tzur YB, Wilson KL, Gruenbaum Y (2006) Nat Rev Mol Cell Biol 7: Malone CJ, Fixsen WD, Horvitz HR, Han M (1999) Development (Cambridge, UK) 126: Hodzic DM, Yeater DB, Bengtsson L, Otto H, Stahl PD (2004) J Biol Chem 279: Dechat T, Vlcek S, Foisner R (2000) J Struct Biol 129: Scherthan H, Weich S, Schwegler H, Heyting C, Harle M, Cremer T (1996) J Cell Biol 134: Starr DA, Fischer JA (2005) BioEssays 27: Sullivan T, Escalante-Alcalde D, Bhatt H, Anver M, Bhat N, Nagashima K, Stewart CL, Burke B (1999) J Cell Biol 147: Hasan S, Güttinger S, Mühlhäusser P, Anderegg F, Bürgler S, Kutay U (2006) FEBS Lett 580: Yuan L, Liu JG, Zhao J, Brundell E, Daneholt B, Höög C (2000) Mol Cell 5: Chikashige Y, Ding DQ, Imai Y, Yamamoto M, Haraguchi T, Hiraoka Y (1997) EMBO J 16: Hagan I, Yanagida M (1995) J Cell Biol 129: Chikashige Y, Tsutsumi C, Yamane M, Okamasa K, Haraguchi T, Hiraoka Y (2006) Cell 125: Worman HJ, Gundersen GG (2006) Trends Cell Biol 16: Tomita K, Cooper JP (2006) Cell 125: Tang X, Jin Y, Cande WZ (2006) J Cell Biol 173: Shimanuki M, Miki F, Ding DQ, Chikashige Y, Hiraoka Y, Horio T, Niwa O (1997) Mol Gen Genet 254: Miki F, Okazaki K, Shimanuki M, Yamamoto A, Hiraoka Y, Niwa O (2002) Mol Biol Cell 13: Miki F, Kurabayashi A, Tange Y, Okazaki K, Shimanuki M, Niwa O (2004) Mol Genet Genomics 270: Fridkin A, Mills E, Margalit A, Neufeld E, Lee KK, Feinstein N, Cohen M, Wilson KL, Gruenbaum Y (2004) Proc Natl Acad Sci USA 101: Padmakumar VC, Abraham S, Braune S, Noegel AA, Tunggal B, Karakesisoglou I, Korenbaum E (2004) Exp Cell Res 295: Trelles-Sticken E, Adelfalk C, Loidl J, Scherthan H (2005) J Cell Biol 170: Parvinen M, Söderström KO (1976) Nature 260: Alsheimer M, Fecher E, Benavente R (1998) J Cell Sci 111: CELL BIOLOGY Schmitt et al. PNAS May 1, 2007 vol. 104 no
CHAPTER 10 : CELL CYCLE AND CELL DIVISION K C MEENA PGT BIOLOGY KVS
 CHAPTER 10 : CELL CYCLE AND CELL DIVISION K C MEENA PGT BIOLOGY KVS Cell cycle It is a series of events that takes place in a cell, leading to the formation of two daughter cells from a single mother cell.
CHAPTER 10 : CELL CYCLE AND CELL DIVISION K C MEENA PGT BIOLOGY KVS Cell cycle It is a series of events that takes place in a cell, leading to the formation of two daughter cells from a single mother cell.
CELL CYCLE AND CELL DIVISION
 1 CH 10 CELL CYCLE & CELL DIVISION CELL CYCLE AND CELL DIVISION Growth and reproduction are characteristics of living cells and organisms. Cell Cycle The sequence of events by which a cell duplicates its
1 CH 10 CELL CYCLE & CELL DIVISION CELL CYCLE AND CELL DIVISION Growth and reproduction are characteristics of living cells and organisms. Cell Cycle The sequence of events by which a cell duplicates its
CLASS XI CHAPTER 10 CELL CYCLE AND CELL DIVISION
 CLASS XI CHAPTER 10 CELL CYCLE AND CELL DIVISION Cell cycle It is a series of events that takes place in a cell, leading to the formation of two daughter cells from a single mother cell. Phases of cell
CLASS XI CHAPTER 10 CELL CYCLE AND CELL DIVISION Cell cycle It is a series of events that takes place in a cell, leading to the formation of two daughter cells from a single mother cell. Phases of cell
Visit For All NCERT solutions, CBSE sample papers, Question papers, Notes for Class 6 to 12. Chapter-10
 Chapter-10 CELL CYCLE AND CELL DIVISION POINTS TO REMEMBER Cell cycle : The sequence of events by which a cell duplicates its genome, synthesis the other constitutents of the cell and eventually divides
Chapter-10 CELL CYCLE AND CELL DIVISION POINTS TO REMEMBER Cell cycle : The sequence of events by which a cell duplicates its genome, synthesis the other constitutents of the cell and eventually divides
Reading Assignments. A. Systems of Cell Division. Lecture Series 5 Cell Cycle & Cell Division
 Lecture Series 5 Cell Cycle & Cell Division Reading Assignments Read Chapter 18 Cell Cycle & Cell Death Read Chapter 19 Cell Division Read Chapter 20 pages 659-672 672 only (Benefits of Sex & Meiosis sections)
Lecture Series 5 Cell Cycle & Cell Division Reading Assignments Read Chapter 18 Cell Cycle & Cell Death Read Chapter 19 Cell Division Read Chapter 20 pages 659-672 672 only (Benefits of Sex & Meiosis sections)
Lecture Series 5 Cell Cycle & Cell Division
 Lecture Series 5 Cell Cycle & Cell Division Reading Assignments Read Chapter 18 Cell Cycle & Cell Death Read Chapter 19 Cell Division Read Chapter 20 pages 659-672 672 only (Benefits of Sex & Meiosis sections)
Lecture Series 5 Cell Cycle & Cell Division Reading Assignments Read Chapter 18 Cell Cycle & Cell Death Read Chapter 19 Cell Division Read Chapter 20 pages 659-672 672 only (Benefits of Sex & Meiosis sections)
Lecture Series 5 Cell Cycle & Cell Division
 Lecture Series 5 Cell Cycle & Cell Division Reading Assignments Read Chapter 18 Cell Cycle & Cell Division Read Chapter 19 pages 651-663 663 only (Benefits of Sex & Meiosis sections these are in Chapter
Lecture Series 5 Cell Cycle & Cell Division Reading Assignments Read Chapter 18 Cell Cycle & Cell Division Read Chapter 19 pages 651-663 663 only (Benefits of Sex & Meiosis sections these are in Chapter
E. Incorrect! At telophase II, cells are nearly completed with meiosis, with no cross-over.
 OAT Biology - Problem Drill 06: Mitosis and Meiosis Question No. 1 of 10 1. During meiosis, cross-over between homologous chromosomes occurs at the end of. Question #01 (A) Anaphase II (B) Metaphase I
OAT Biology - Problem Drill 06: Mitosis and Meiosis Question No. 1 of 10 1. During meiosis, cross-over between homologous chromosomes occurs at the end of. Question #01 (A) Anaphase II (B) Metaphase I
The interphase is further divided into three phases, which are as follows:
 Cell Division Phases of Cell Cycle The cell cycle is divided into two basic phases: a. Interphase: The phase between subsequent cell divisions is called the interphase. The interphase lasts for more than
Cell Division Phases of Cell Cycle The cell cycle is divided into two basic phases: a. Interphase: The phase between subsequent cell divisions is called the interphase. The interphase lasts for more than
The Gametic Cell Cycle
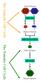 The Gametic Cell Cycle Diploid Parents MEIOSIS Haploid Gametes FERTILIZATION The Somatic Cell Cycle Diploid Zygote MITOSIS Diploid organism Activities of meiosis that differ from mitosis? Life cycle of
The Gametic Cell Cycle Diploid Parents MEIOSIS Haploid Gametes FERTILIZATION The Somatic Cell Cycle Diploid Zygote MITOSIS Diploid organism Activities of meiosis that differ from mitosis? Life cycle of
Human biology Laboratory. Cell division. Lecturer Maysam A Mezher
 Human biology Laboratory Cell division Lecturer Maysam A Mezher CHROMOSOME STRUCTURE 1. During nuclear division, the DNA (as chromatin) in a Eukaryotic cell's nucleus is coiled into very tight compact
Human biology Laboratory Cell division Lecturer Maysam A Mezher CHROMOSOME STRUCTURE 1. During nuclear division, the DNA (as chromatin) in a Eukaryotic cell's nucleus is coiled into very tight compact
The SUN Rises on Meiotic Chromosome Dynamics
 The SUN Rises on Meiotic Chromosome Dynamics Yasushi Hiraoka 1,2, * and Abby F. Dernburg 3,4, * 1 Graduate School of Frontier Biosciences, Osaka University, 1-3 Yamadaoka, Suita 565-0871, Japan 2 Kobe
The SUN Rises on Meiotic Chromosome Dynamics Yasushi Hiraoka 1,2, * and Abby F. Dernburg 3,4, * 1 Graduate School of Frontier Biosciences, Osaka University, 1-3 Yamadaoka, Suita 565-0871, Japan 2 Kobe
ASSAY OF CHROMOSOME MOVEMENT AND PAIRING DURING MEIOSIS IN SACCHAROMYCES CEREVISIAE USING LIVE CELL IMAGING
 ASSAY OF CHROMOSOME MOVEMENT AND PAIRING DURING MEIOSIS IN SACCHAROMYCES CEREVISIAE USING LIVE CELL IMAGING James McGehee Abstract Meiosis is a specialized form of cell division, in which homologous chromosomes
ASSAY OF CHROMOSOME MOVEMENT AND PAIRING DURING MEIOSIS IN SACCHAROMYCES CEREVISIAE USING LIVE CELL IMAGING James McGehee Abstract Meiosis is a specialized form of cell division, in which homologous chromosomes
NCERT. not to be published CHAPTER 10 CELL CYCLE AND CELL DIVISION 10.1 CELL CYCLE 162 BIOLOGY
 162 BIOLOGY 10.1 Cell Cycle 10.2 M Phase 10.3 Significance of Mitosis 10.4 Meiosis 10.5 Significance of Meiosis CHAPTER 10 CELL CYCLE AND CELL DIVISION Are you aware that all organisms, even the largest,
162 BIOLOGY 10.1 Cell Cycle 10.2 M Phase 10.3 Significance of Mitosis 10.4 Meiosis 10.5 Significance of Meiosis CHAPTER 10 CELL CYCLE AND CELL DIVISION Are you aware that all organisms, even the largest,
CELL GROWTH AND DIVISION. Chapter 10
 CELL GROWTH AND DIVISION Chapter 10 Cell division = The formation of 2 daughter cells from a single parent cell Increases ratio of surface area to volume for each cell Allows for more efficient exchange
CELL GROWTH AND DIVISION Chapter 10 Cell division = The formation of 2 daughter cells from a single parent cell Increases ratio of surface area to volume for each cell Allows for more efficient exchange
Sexual Reproduction and Meiosis. Chapter 11
 Sexual Reproduction and Meiosis Chapter 11 1 Sexual life cycle Made up of meiosis and fertilization Diploid cells Somatic cells of adults have 2 sets of chromosomes Haploid cells Gametes (egg and sperm)
Sexual Reproduction and Meiosis Chapter 11 1 Sexual life cycle Made up of meiosis and fertilization Diploid cells Somatic cells of adults have 2 sets of chromosomes Haploid cells Gametes (egg and sperm)
2:1 Chromosomes DNA Genes Chromatin Chromosomes CHROMATIN: nuclear material in non-dividing cell, composed of DNA/protein in thin uncoiled strands
 Human Heredity Chapter 2 Chromosomes, Mitosis, and Meiosis 2:1 Chromosomes DNA Genes Chromatin Chromosomes CHROMATIN: nuclear material in non-dividing cell, composed of DNA/protein in thin uncoiled strands
Human Heredity Chapter 2 Chromosomes, Mitosis, and Meiosis 2:1 Chromosomes DNA Genes Chromatin Chromosomes CHROMATIN: nuclear material in non-dividing cell, composed of DNA/protein in thin uncoiled strands
Typical Life Cycle of Algae and Fungi. 5 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
 Module 3B Meiosis and Sexual Life Cycles In this module, we will examine a second type of cell division used by eukaryotic cells called meiosis. In addition, we will see how the 2 types of eukaryotic cell
Module 3B Meiosis and Sexual Life Cycles In this module, we will examine a second type of cell division used by eukaryotic cells called meiosis. In addition, we will see how the 2 types of eukaryotic cell
Cell Division. Mitosis
 Cell division consists of two phases, nuclear division followed by cytokinesis. Nuclear division divides the genetic material in the nucleus, while cytokinesis divides the cytoplasm. There are two kinds
Cell division consists of two phases, nuclear division followed by cytokinesis. Nuclear division divides the genetic material in the nucleus, while cytokinesis divides the cytoplasm. There are two kinds
Biology Unit 6 Chromosomes and Mitosis
 Biology Unit 6 Chromosomes and Mitosis 6:1 Chromosomes DNA GENES CHROMATIN/CHROMOSOMES CHROMOSOMES/CHROMATIN are made of units called GENES. GENES are made of a compound called deoxyribonucleic acid or
Biology Unit 6 Chromosomes and Mitosis 6:1 Chromosomes DNA GENES CHROMATIN/CHROMOSOMES CHROMOSOMES/CHROMATIN are made of units called GENES. GENES are made of a compound called deoxyribonucleic acid or
GENERAL SAFETY: Follow your teacher s directions. Do not work in the laboratory without your teacher s supervision.
 Name: Bio AP Lab: Cell Division B: Mitosis & Meiosis (Modified from AP Biology Investigative Labs) BACKGROUND: One of the characteristics of living things is the ability to replicate and pass on genetic
Name: Bio AP Lab: Cell Division B: Mitosis & Meiosis (Modified from AP Biology Investigative Labs) BACKGROUND: One of the characteristics of living things is the ability to replicate and pass on genetic
 Meiosis http://biology.tutorvista.com/cell/meiosis.html MEIOSIS It is a type of cell division in which the chromosome number is halved from the diploid number (2n) to a haploid number (n). Like mitosis
Meiosis http://biology.tutorvista.com/cell/meiosis.html MEIOSIS It is a type of cell division in which the chromosome number is halved from the diploid number (2n) to a haploid number (n). Like mitosis
Meiosis. Section 8-3
 Meiosis Section 8-3 Meiosis process of nuclear division that reduces the number of chromosomes in new cells to half the number in the original cell For example, in humans, meiosis produces haploid reproductive
Meiosis Section 8-3 Meiosis process of nuclear division that reduces the number of chromosomes in new cells to half the number in the original cell For example, in humans, meiosis produces haploid reproductive
Chapter 2: Chromosomes and cellular reproduction
 Chapter 2: Chromosomes and cellular reproduction I. Contrast between prokaryotes and eukaryotes. See Figure 2.1 Nucleus absent Small diameter 1 to 10 µm Genome usually 1 circular molecule Small genome;
Chapter 2: Chromosomes and cellular reproduction I. Contrast between prokaryotes and eukaryotes. See Figure 2.1 Nucleus absent Small diameter 1 to 10 µm Genome usually 1 circular molecule Small genome;
A bouquet of chromosomes
 Commentary 4025 A bouquet of chromosomes Lisa Harper, Inna Golubovskaya and W. Zacheus Cande* Department of Molecular and Cell Biology, University of California Berkeley, 345 LSA 3200, Berkeley, CA 94720-3200,
Commentary 4025 A bouquet of chromosomes Lisa Harper, Inna Golubovskaya and W. Zacheus Cande* Department of Molecular and Cell Biology, University of California Berkeley, 345 LSA 3200, Berkeley, CA 94720-3200,
Meiosis. Bởi: OpenStaxCollege
 Meiosis Bởi: OpenStaxCollege Sexual reproduction requires fertilization, a union of two cells from two individual organisms. If those two cells each contain one set of chromosomes, then the resulting cell
Meiosis Bởi: OpenStaxCollege Sexual reproduction requires fertilization, a union of two cells from two individual organisms. If those two cells each contain one set of chromosomes, then the resulting cell
Biology 067 Section 14 Cell Division. A. Definitions:
 Biology 067 Section 14 Cell Division A. Definitions: In a human cell, a nucleus holds all the chromatin that condenses to form chromosomes when cells divide every cell in the body has the same set of chromosomes
Biology 067 Section 14 Cell Division A. Definitions: In a human cell, a nucleus holds all the chromatin that condenses to form chromosomes when cells divide every cell in the body has the same set of chromosomes
Meiosis Production of Chromosome Hybrids & Gametes. Packet #29
 Meiosis Production of Chromosome Hybrids & Gametes Packet #29 Introduction Meiosis, discovered in 1883, was first observed through the fertilized egg and individual gametes, sperm and unfertilized egg,
Meiosis Production of Chromosome Hybrids & Gametes Packet #29 Introduction Meiosis, discovered in 1883, was first observed through the fertilized egg and individual gametes, sperm and unfertilized egg,
8.8 Growth factors signal the cell cycle control system
 The Cellular Basis of Reproduction and Inheritance : part II PowerPoint Lectures for Campbell Biology: Concepts & Connections, Seventh Edition Reece, Taylor, Simon, and Dickey Biology 1408 Dr. Chris Doumen
The Cellular Basis of Reproduction and Inheritance : part II PowerPoint Lectures for Campbell Biology: Concepts & Connections, Seventh Edition Reece, Taylor, Simon, and Dickey Biology 1408 Dr. Chris Doumen
Mitosis and Meiosis Cell growth and division
 LIMITS TO CELL GROWTH Mitosis and Meiosis Cell growth and division The larger the cell, the more trouble the cell has moving nutrients and waste across the cell membrane. LIMITS TO CELL GROWTH 1. DNA/information
LIMITS TO CELL GROWTH Mitosis and Meiosis Cell growth and division The larger the cell, the more trouble the cell has moving nutrients and waste across the cell membrane. LIMITS TO CELL GROWTH 1. DNA/information
Meiosis * OpenStax. This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0.
 OpenStax-CNX module: m45466 1 Meiosis * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be able to: Abstract
OpenStax-CNX module: m45466 1 Meiosis * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be able to: Abstract
Fertilization of sperm and egg produces offspring
 In sexual reproduction Fertilization of sperm and egg produces offspring In asexual reproduction Offspring are produced by a single parent, without the participation of sperm and egg CONNECTIONS BETWEEN
In sexual reproduction Fertilization of sperm and egg produces offspring In asexual reproduction Offspring are produced by a single parent, without the participation of sperm and egg CONNECTIONS BETWEEN
Biology. Chapter 10 Cell Reproduction. I. Chromosomes
 Biology Chapter 10 Cell Reproduction I. Chromosomes Long thin molecules that store genetic information. A. Chromosome Structure 1. Rod shaped structure composed of DNA and protein. 2. DNA is wrapped around
Biology Chapter 10 Cell Reproduction I. Chromosomes Long thin molecules that store genetic information. A. Chromosome Structure 1. Rod shaped structure composed of DNA and protein. 2. DNA is wrapped around
Mitosis and. Meiosis. Presented by Kesler Science
 Mitosis and Meiosis Presented by Kesler Science Essential Questions: 1. What are mitosis and meiosis? 2. What occurs at different phases in cell division? 3. How are mitosis and meiosis similar and different?
Mitosis and Meiosis Presented by Kesler Science Essential Questions: 1. What are mitosis and meiosis? 2. What occurs at different phases in cell division? 3. How are mitosis and meiosis similar and different?
MEIOSIS LAB INTRODUCTION PART I: SIMULATION OF MEIOSIS EVOLUTION. Activity #9
 AP BIOLOGY EVOLUTION Unit 1 Part 7 Chapter 13 Activity #9 NAME DATE PERIOD MEIOSIS LAB INTRODUCTION Meiosis involves two successive nuclear divisions that produce four haploid cells. Meiosis I is the reduction
AP BIOLOGY EVOLUTION Unit 1 Part 7 Chapter 13 Activity #9 NAME DATE PERIOD MEIOSIS LAB INTRODUCTION Meiosis involves two successive nuclear divisions that produce four haploid cells. Meiosis I is the reduction
Cellular Division. copyright cmassengale
 Cellular Division 1 Cell Division All cells are derived from pre- existing cells New cells are produced for growth and to replace damaged or old cells Differs in prokaryotes (bacteria) and eukaryotes (protists,
Cellular Division 1 Cell Division All cells are derived from pre- existing cells New cells are produced for growth and to replace damaged or old cells Differs in prokaryotes (bacteria) and eukaryotes (protists,
Topic 8 Mitosis & Meiosis Ch.12 & 13. The Eukaryotic Genome. The Eukaryotic Genome. The Eukaryotic Genome
 Topic 8 Mitosis & Meiosis Ch.12 & 13 The Eukaryotic Genome pp. 244-245,268-269 Genome All of the genes in a cell. Eukaryotic cells contain their DNA in long linear pieces. In prokaryotic cells, there is
Topic 8 Mitosis & Meiosis Ch.12 & 13 The Eukaryotic Genome pp. 244-245,268-269 Genome All of the genes in a cell. Eukaryotic cells contain their DNA in long linear pieces. In prokaryotic cells, there is
CHAPTER 10 CELL CYCLE AND CELL DIVISION MULTIPLE CHOICE QUESTIONS
 CELL CYCLE AND CELL DIVISION 49 49 CHAPTER 10 CELL CYCLE AND CELL DIVISION MULTIPLE CHOICE QUESTIONS 1. Meiosis results in a. Production of gametes b. Reduction in the number of chromosomes c. Introduction
CELL CYCLE AND CELL DIVISION 49 49 CHAPTER 10 CELL CYCLE AND CELL DIVISION MULTIPLE CHOICE QUESTIONS 1. Meiosis results in a. Production of gametes b. Reduction in the number of chromosomes c. Introduction
Unit 6 Test: The Cell Cycle
 Name Date Class Mrs. Knight Biology EHS Unit 6 Test: The Cell Cycle 1. What are the four main stages of the cell cycle (correct order)? A. G 1, S, G 0, M C. G 2, S, G 1, M B. G 1, S, G 2, M D. M, G 2,
Name Date Class Mrs. Knight Biology EHS Unit 6 Test: The Cell Cycle 1. What are the four main stages of the cell cycle (correct order)? A. G 1, S, G 0, M C. G 2, S, G 1, M B. G 1, S, G 2, M D. M, G 2,
ACCELERATE ITS BIOCHEMICAL PROCESSES WHICH WERE SLOWED DOWN BY MITOSIS. THE LENGTH OF THE G1 PHASE CREATES THE DIFFERENCE BETWEEN FAST DIVIDING
 CHAPTER 1: OVERVIEW OF THE CELL CYCLE THE THREE STAGES OF INTERPHASE: INTERPHASE BEFORE A CELL CAN ENTER CELL DIVISION, IT NEEDS TO PREPARE ITSELF BY REPLICATING ITS GENETIC INFORMATION AND ALL OF THE
CHAPTER 1: OVERVIEW OF THE CELL CYCLE THE THREE STAGES OF INTERPHASE: INTERPHASE BEFORE A CELL CAN ENTER CELL DIVISION, IT NEEDS TO PREPARE ITSELF BY REPLICATING ITS GENETIC INFORMATION AND ALL OF THE
Meiosis: M-Phase part 2. How is meiosis different from mitosis? Some terms: Some terms: Some terms:
 M-Phase part 2 Meiosis and Cytokinesis Meiosis: A special type of cell division that produces gametes. Cell division that occurs in sex organs. 2 successive nuclear divisions Genetic mistakes (gene and
M-Phase part 2 Meiosis and Cytokinesis Meiosis: A special type of cell division that produces gametes. Cell division that occurs in sex organs. 2 successive nuclear divisions Genetic mistakes (gene and
Cell Division. Mitosis 11/8/2016
 Cell division consists of two phases, nuclear division followed by cytokinesis. Nuclear division divides the genetic material in the nucleus, while cytokinesis divides the cytoplasm. There are two kinds
Cell division consists of two phases, nuclear division followed by cytokinesis. Nuclear division divides the genetic material in the nucleus, while cytokinesis divides the cytoplasm. There are two kinds
Mitosis, development, regeneration and cell differentiation
 Mitosis, development, regeneration and cell differentiation Mitosis is a type of cell division by binary fission (splitting in two) which occurs in certain eukaryotic cells. Mitosis generates new body
Mitosis, development, regeneration and cell differentiation Mitosis is a type of cell division by binary fission (splitting in two) which occurs in certain eukaryotic cells. Mitosis generates new body
Meiosis, Sexual Reproduction, & Genetic Variability
 Meiosis, Sexual Reproduction, & Genetic Variability Teachers Guide NARRATION FOR MEIOSIS, SEXUAL REPRODUCTION, AND GENETIC VARIABILITY Since the members of no species, even California redwoods or giant
Meiosis, Sexual Reproduction, & Genetic Variability Teachers Guide NARRATION FOR MEIOSIS, SEXUAL REPRODUCTION, AND GENETIC VARIABILITY Since the members of no species, even California redwoods or giant
BIOLOGY 111. CHAPTER 5: Chromosomes and Inheritance
 BIOLOGY 111 CHAPTER 5: Chromosomes and Inheritance Chromosomes and Inheritance Learning Outcomes 5.1 Differentiate between sexual and asexual reproduction in terms of the genetic variation of the offspring.
BIOLOGY 111 CHAPTER 5: Chromosomes and Inheritance Chromosomes and Inheritance Learning Outcomes 5.1 Differentiate between sexual and asexual reproduction in terms of the genetic variation of the offspring.
The Microscopic Observation of Mitosis in Plant and Animal Cells
 The Microscopic Observation of Mitosis in Plant and Animal Cells Prelab Assignment Before coming to lab, read carefully the introduction and the procedures for each part of the experiment, and then answer
The Microscopic Observation of Mitosis in Plant and Animal Cells Prelab Assignment Before coming to lab, read carefully the introduction and the procedures for each part of the experiment, and then answer
CELL REPRODUCTION NOTES
 CELL REPRODUCTION NOTES CELL GROWTH AND DIVISION The adult human body produces roughly cells every day. WHY DO CELLS REPRODUCE? So that the organism can and As multicellular organisms grow larger, its
CELL REPRODUCTION NOTES CELL GROWTH AND DIVISION The adult human body produces roughly cells every day. WHY DO CELLS REPRODUCE? So that the organism can and As multicellular organisms grow larger, its
3.a.2- Cell Cycle and Meiosis
 Big Idea 3: Living systems store, retrieve, transmit and respond to information essential to life processes. 3.a.2- Cell Cycle and Meiosis EU 3.A: Heritable information provides for continuity of life.
Big Idea 3: Living systems store, retrieve, transmit and respond to information essential to life processes. 3.a.2- Cell Cycle and Meiosis EU 3.A: Heritable information provides for continuity of life.
2. Which of the following are NOT prokaryotes? A) eubacteria B) archaea C) viruses D) ancient bacteria
 1. Which of the following statements is FALSE? A) Errors in chromosome separation are rarely a problem for an organism. B) Errors in chromosome separation can result in a miscarriage. C) Errors in chromosome
1. Which of the following statements is FALSE? A) Errors in chromosome separation are rarely a problem for an organism. B) Errors in chromosome separation can result in a miscarriage. C) Errors in chromosome
Cell Reproduction Review
 Name Date Period Cell Reproduction Review Explain what is occurring in each part of the cell cycle --- G 0, G1, S, G2, and M. 1 CELL DIVISION Label all parts of each cell in the cell cycle and explain
Name Date Period Cell Reproduction Review Explain what is occurring in each part of the cell cycle --- G 0, G1, S, G2, and M. 1 CELL DIVISION Label all parts of each cell in the cell cycle and explain
Cell Division (Meiosis)
 Cell Division (Meiosis) Meiosis The form of cell division by which gametes, with half the number of chromosomes, are produced. Diploid (2n) haploid (n) Meiosis is sexual reproduction. Two divisions (meiosis
Cell Division (Meiosis) Meiosis The form of cell division by which gametes, with half the number of chromosomes, are produced. Diploid (2n) haploid (n) Meiosis is sexual reproduction. Two divisions (meiosis
The Cell Cycles Mitosis and Meiosis. Essential question: How do cells reproduce and why?
 The Cell Cycles Mitosis and Meiosis Essential question: How do cells reproduce and why? Objectives Section 10.1 Explain why cells divide in terms of growth and cell size Review - Types of Cell Division
The Cell Cycles Mitosis and Meiosis Essential question: How do cells reproduce and why? Objectives Section 10.1 Explain why cells divide in terms of growth and cell size Review - Types of Cell Division
Biology, 7e (Campbell) Chapter 13: Meiosis and Sexual Life Cycles
 Biology, 7e (Campbell) Chapter 13: Meiosis and Sexual Life Cycles Chapter Questions 1) What is a genome? A) the complete complement of an organism's genes B) a specific sequence of polypeptides within
Biology, 7e (Campbell) Chapter 13: Meiosis and Sexual Life Cycles Chapter Questions 1) What is a genome? A) the complete complement of an organism's genes B) a specific sequence of polypeptides within
You have body cells and gametes Body cells are known as somatic cells. Germ cells develop into gametes or sex cells. Germ cells are located in the
 MEIOSIS You have body cells and gametes Body cells are known as somatic cells. Germ cells develop into gametes or sex cells. Germ cells are located in the ovaries and testes. Gametes are sex cells: egg
MEIOSIS You have body cells and gametes Body cells are known as somatic cells. Germ cells develop into gametes or sex cells. Germ cells are located in the ovaries and testes. Gametes are sex cells: egg
Human Biology Chapter 13.4: Meiosis and Genetic Variation
 OpenStax-CNX module: m58013 1 Human Biology Chapter 13.4: Meiosis and Genetic Variation Willy Cushwa Based on Meiosis by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons
OpenStax-CNX module: m58013 1 Human Biology Chapter 13.4: Meiosis and Genetic Variation Willy Cushwa Based on Meiosis by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons
VIII. A Time-Course Study of the Fusion and Division of the Spindle Pole
 Published Online: 1 March, 1978 Supp Info: http://doi.org/10.1083/jcb.76.3.761 Downloaded from jcb.rupress.org on June 30, 2018 MEIOSIS IN COPRINUS VIII. A Time-Course Study of the Fusion and Division
Published Online: 1 March, 1978 Supp Info: http://doi.org/10.1083/jcb.76.3.761 Downloaded from jcb.rupress.org on June 30, 2018 MEIOSIS IN COPRINUS VIII. A Time-Course Study of the Fusion and Division
CHAPTER 10 CELL CYCLE AND CELL DIVISION
 162 BIOLOGY CHAPTER 10 CELL CYCLE AND CELL DIVISION 10.1 Cell Cycle 10.2 M Phase 10.3 Significance of Mitosis 10.4 Meiosis 10.5 Significance of Meiosis Are you aware that all organisms, even the largest,
162 BIOLOGY CHAPTER 10 CELL CYCLE AND CELL DIVISION 10.1 Cell Cycle 10.2 M Phase 10.3 Significance of Mitosis 10.4 Meiosis 10.5 Significance of Meiosis Are you aware that all organisms, even the largest,
AP Biology - Cell cycle / division
 AP Biology - Cell cycle / division Quiz Directions 1. During which stage does DNA replication occur? A. Prophase B. Metaphase C. Anaphase D. none of these 2. At what phase in the cell cycle does DNA replication
AP Biology - Cell cycle / division Quiz Directions 1. During which stage does DNA replication occur? A. Prophase B. Metaphase C. Anaphase D. none of these 2. At what phase in the cell cycle does DNA replication
Ladies and Gentlemen.. The King of Rock and Roll
 Ladies and Gentlemen.. The King of Rock and Roll Learning Objectives: The student is able to construct an explanation, using visual representations or narratives, as to how DNA in chromosomes is transmitted
Ladies and Gentlemen.. The King of Rock and Roll Learning Objectives: The student is able to construct an explanation, using visual representations or narratives, as to how DNA in chromosomes is transmitted
Numerous studies in the past years
 Nucleus 6:2, 93--101; March/April 2015; Published with license by Taylor & Francis EXTRA VIEW Structural and functional adaptations of the mammalian nuclear envelope to meet the meiotic requirements Jana
Nucleus 6:2, 93--101; March/April 2015; Published with license by Taylor & Francis EXTRA VIEW Structural and functional adaptations of the mammalian nuclear envelope to meet the meiotic requirements Jana
Chromosome Chr Duplica Duplic t a ion Pixley
 Chromosome Duplication Pixley Figure 4-6 Molecular Biology of the Cell ( Garland Science 2008) Figure 4-72 Molecular Biology of the Cell ( Garland Science 2008) Interphase During mitosis (cell division),
Chromosome Duplication Pixley Figure 4-6 Molecular Biology of the Cell ( Garland Science 2008) Figure 4-72 Molecular Biology of the Cell ( Garland Science 2008) Interphase During mitosis (cell division),
gametes Gametes somatic cells diploid (2n) haploid (n)
 Overview of Meiosis Meiosis is a form of cell division that leads to the production of gametes. Gametes: egg cells and sperm cells (reproductive) -contain half the number of chromosomes of an adult body
Overview of Meiosis Meiosis is a form of cell division that leads to the production of gametes. Gametes: egg cells and sperm cells (reproductive) -contain half the number of chromosomes of an adult body
Chapter 13 Meiosis and Sexual Reproduction
 Biology 110 Sec. 11 J. Greg Doheny Chapter 13 Meiosis and Sexual Reproduction Quiz Questions: 1. What word do you use to describe a chromosome or gene allele that we inherit from our Mother? From our Father?
Biology 110 Sec. 11 J. Greg Doheny Chapter 13 Meiosis and Sexual Reproduction Quiz Questions: 1. What word do you use to describe a chromosome or gene allele that we inherit from our Mother? From our Father?
Ch. 13 Meiosis & Sexual Life Cycles
 Introduction Ch. 13 Meiosis & Sexual Life Cycles 2004-05 Living organisms are distinguished by their ability to reproduce their own kind. -Offspring resemble their parents more than they do less closely
Introduction Ch. 13 Meiosis & Sexual Life Cycles 2004-05 Living organisms are distinguished by their ability to reproduce their own kind. -Offspring resemble their parents more than they do less closely
Class XI Chapter 10 Cell Cycle and Cell Division Biology
 Question 1: What is the average cell cycle span for a mammalian cell? The average cell cycle span for a mammalian cell is approximately 24 hours. Question 2: Distinguish cytokinesis from karyokinesis.
Question 1: What is the average cell cycle span for a mammalian cell? The average cell cycle span for a mammalian cell is approximately 24 hours. Question 2: Distinguish cytokinesis from karyokinesis.
Chapter 8 Lectures by Gregory Ahearn University of North Florida
 Chapter 8 The Continuity of Life: How Cells Reproduce Lectures by Gregory Ahearn University of North Florida Copyright 2009 Pearson Education, Inc. 8.1 Why Do Cells Divide? Cells reproduce by cell division.
Chapter 8 The Continuity of Life: How Cells Reproduce Lectures by Gregory Ahearn University of North Florida Copyright 2009 Pearson Education, Inc. 8.1 Why Do Cells Divide? Cells reproduce by cell division.
LAB 8 EUKARYOTIC CELL DIVISION: MITOSIS AND MEIOSIS
 LAB 8 EUKARYOTIC CELL DIVISION: MITOSIS AND MEIOSIS Name: Date: INTRODUCTION BINARY FISSION: Prokaryotic cells (bacteria) reproduce asexually by binary fission. Bacterial cells have a single circular chromosome,
LAB 8 EUKARYOTIC CELL DIVISION: MITOSIS AND MEIOSIS Name: Date: INTRODUCTION BINARY FISSION: Prokaryotic cells (bacteria) reproduce asexually by binary fission. Bacterial cells have a single circular chromosome,
Asexual vs. Sexual. Biology 3201 Unit II Reproduction How Reproductive Cells are Produced. two parents offspring is unique
 Biology 3201 Unit II Reproduction 14.2 How Reproductive Cells are Produced Asexual vs single parent offspring identical to parent parent passes on ALL its genes results in a clone Sexual two parents offspring
Biology 3201 Unit II Reproduction 14.2 How Reproductive Cells are Produced Asexual vs single parent offspring identical to parent parent passes on ALL its genes results in a clone Sexual two parents offspring
Mitosis vs Meiosis. Mitosis and Meiosis -- Internet Tutorial
 Mitosis and Meiosis -- Internet Tutorial In this internet lesson, you will review the steps of mitosis and meiosis and view video simulations of cell division. Mitosis: An Interactive Animation (http://www.cellsalive.com/mitosis.htm)
Mitosis and Meiosis -- Internet Tutorial In this internet lesson, you will review the steps of mitosis and meiosis and view video simulations of cell division. Mitosis: An Interactive Animation (http://www.cellsalive.com/mitosis.htm)
Meiosis produces haploid gametes.
 Section 1: produces haploid gametes. K What I Know W What I Want to Find Out L What I Learned Essential Questions How does the reduction in chromosome number occur during meiosis? What are the stages of
Section 1: produces haploid gametes. K What I Know W What I Want to Find Out L What I Learned Essential Questions How does the reduction in chromosome number occur during meiosis? What are the stages of
The Cell Cycle and Cell Division
 The Cell Cycle and Cell Division «The cell cycle is a regular pattern of growth, DNA replication, and cell division. The cell cycle has four main stages. «The main stages of the cell cycle are G1 (gap
The Cell Cycle and Cell Division «The cell cycle is a regular pattern of growth, DNA replication, and cell division. The cell cycle has four main stages. «The main stages of the cell cycle are G1 (gap
Overview of Genetic Organization and Scale
 CHAPTER ONE Overview of Genetic Organization and Scale The genetic material is a molecule called deoxyribonucleic acid (DNA). Each chromosome contains a single long strand of DNA that encodes the information
CHAPTER ONE Overview of Genetic Organization and Scale The genetic material is a molecule called deoxyribonucleic acid (DNA). Each chromosome contains a single long strand of DNA that encodes the information
BIOLOGY CLASS 10 Chapter 2 Cell cycle, cell division and structure of chromosomes
 BIOLOGY CLASS 10 Chapter 2 Cell cycle, cell division and structure of chromosomes 1) Cell division is an important process in all living things. State any four reasons to support your answer. New cells
BIOLOGY CLASS 10 Chapter 2 Cell cycle, cell division and structure of chromosomes 1) Cell division is an important process in all living things. State any four reasons to support your answer. New cells
How did that single cell develop into a body with more than a trillion cells?
 CELL DIVISION Cell division is a process which leads to cell multiplication. It occurs in both plants and animals. Original cells which undergo division are known as parent cells and the new on ones resulting
CELL DIVISION Cell division is a process which leads to cell multiplication. It occurs in both plants and animals. Original cells which undergo division are known as parent cells and the new on ones resulting
Chapter 13: Meiosis and Sexual Life Cycles Overview: Hereditary Similarity and Variation
 Chapter 13: Meiosis and Sexual Life Cycles Overview: Hereditary Similarity and Variation Living organisms Are distinguished by their ability to reproduce their own kind Biology, 7 th Edition Neil Campbell
Chapter 13: Meiosis and Sexual Life Cycles Overview: Hereditary Similarity and Variation Living organisms Are distinguished by their ability to reproduce their own kind Biology, 7 th Edition Neil Campbell
Key Concepts. n Cell Cycle. n Interphase. n Mitosis. n Cytokinesis
 The Cell Cycle B-2.6: Summarize the characteristics of the cell cycle: interphase (G 1, S, G 2 ); the phases of mitosis (prophase, metaphase, anaphase, telophase); and plant and animal cytokinesis. Key
The Cell Cycle B-2.6: Summarize the characteristics of the cell cycle: interphase (G 1, S, G 2 ); the phases of mitosis (prophase, metaphase, anaphase, telophase); and plant and animal cytokinesis. Key
Rap1-independent telomere attachment and bouquet formation in mammalian meiosis
 Rap1-independent telomere attachment and bouquet formation in mammalian meiosis Chromosoma Biology of the Nucleus ISSN 0009-5915 Volume 120 Number 2 Chromosoma (2011) 120:151-157 DOI 10.1007/ s00412-010-0295-4
Rap1-independent telomere attachment and bouquet formation in mammalian meiosis Chromosoma Biology of the Nucleus ISSN 0009-5915 Volume 120 Number 2 Chromosoma (2011) 120:151-157 DOI 10.1007/ s00412-010-0295-4
Meiosis and Sexual Life Cycles
 Chapter 13 Meiosis and Sexual Life Cycles Lecture Outline Overview Living organisms are distinguished by their ability to reproduce their own kind. Offspring resemble their parents more than they do less
Chapter 13 Meiosis and Sexual Life Cycles Lecture Outline Overview Living organisms are distinguished by their ability to reproduce their own kind. Offspring resemble their parents more than they do less
Lab IV-3. Investigating Cell Division EQUIPMENT AND MATERIALS BACKGROUND
 Investigating Cell Division Lab IV-3 EQUIPMENT AND MATERIALS You ll need the following items to complete this lab session. (The standard kit for this book, available from www.thehomescientist.com, includes
Investigating Cell Division Lab IV-3 EQUIPMENT AND MATERIALS You ll need the following items to complete this lab session. (The standard kit for this book, available from www.thehomescientist.com, includes
Sexual Reproduction ( Cell Division ) - Chromosome # s
 Sexual Reproduction ( Cell Division ) - Chromosome # s somatic cells: all the cells in the body except for specialized sex cells each somatic cell has a specific # of chromosomes - ( humans have 46, 23
Sexual Reproduction ( Cell Division ) - Chromosome # s somatic cells: all the cells in the body except for specialized sex cells each somatic cell has a specific # of chromosomes - ( humans have 46, 23
Anaphase, Telophase. Animal cells divide their cytoplasm by forming? Cleavage furrow. Bacteria, Paramecium, Amoeba, etc. reproduce by...
 The 4 phases of mitosis Animal cells divide their cytoplasm by forming? Bacteria, Paramecium, Amoeba, etc. reproduce by... Cell which after division is identical to the original is called a Prophase, Metaphase,
The 4 phases of mitosis Animal cells divide their cytoplasm by forming? Bacteria, Paramecium, Amoeba, etc. reproduce by... Cell which after division is identical to the original is called a Prophase, Metaphase,
Mitosis and Meiosis for AP Biology
 Mitosis and Meiosis for AP Biology by Mark Anestis Practice problems for these concepts can be found at : Cell Division Review Questions for AP Biology Mitosis During mitosis, the fourth stage of the cell
Mitosis and Meiosis for AP Biology by Mark Anestis Practice problems for these concepts can be found at : Cell Division Review Questions for AP Biology Mitosis During mitosis, the fourth stage of the cell
Honors Biology Test Chapter 8 Mitosis and Meiosis
 Honors Biology Test Chapter 8 Mitosis and Meiosis 1. In mitosis, if a parent cell has 16 chromosomes, each daughter cell will have how many chromosomes? a. 64 b. 32 c. 16 d. 8 e. 4 2. Chromatids that are
Honors Biology Test Chapter 8 Mitosis and Meiosis 1. In mitosis, if a parent cell has 16 chromosomes, each daughter cell will have how many chromosomes? a. 64 b. 32 c. 16 d. 8 e. 4 2. Chromatids that are
16 The Cell Cycle. Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization
 The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions.
 5.1 10.1 The Cell Cell Growth Cycle KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. 5.1 10.1 The Cell Cell Growth Cycle Why must cells divide? Growth and Repair -
5.1 10.1 The Cell Cell Growth Cycle KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. 5.1 10.1 The Cell Cell Growth Cycle Why must cells divide? Growth and Repair -
Class XI Chapter 10 Cell Cycle and Cell Division Biology
 Question 1: What is the average cell cycle span for a mammalian cell? The average cell cycle span for a mammalian cell is approximately 24 hours. Question 2: Distinguish cytokinesis from karyokinesis.
Question 1: What is the average cell cycle span for a mammalian cell? The average cell cycle span for a mammalian cell is approximately 24 hours. Question 2: Distinguish cytokinesis from karyokinesis.
Unit 5: Cell Division and Development Guided Reading Questions (45 pts total)
 Name: AP Biology Biology, Campbell and Reece, 7th Edition Adapted from chapter reading guides originally created by Lynn Miriello Chapter 12 The Cell Cycle Unit 5: Cell Division and Development Guided
Name: AP Biology Biology, Campbell and Reece, 7th Edition Adapted from chapter reading guides originally created by Lynn Miriello Chapter 12 The Cell Cycle Unit 5: Cell Division and Development Guided
MEIOSIS LAB INTRODUCTION PART I: MEIOSIS
 MEIOSIS LAB INTRODUCTION Meiosis involves two successive nuclear divisions that produce four haploid cells. Meiosis I is the reduction division. It is this first division that reduces the chromosome number
MEIOSIS LAB INTRODUCTION Meiosis involves two successive nuclear divisions that produce four haploid cells. Meiosis I is the reduction division. It is this first division that reduces the chromosome number
Meiosis and Sexual Life Cycles
 CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 10 Meiosis and Sexual Life Cycles Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge, Simon Fraser University SECOND EDITION
CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 10 Meiosis and Sexual Life Cycles Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge, Simon Fraser University SECOND EDITION
CELL BIOLOGY - CLUTCH CH MEIOSIS AND SEXUAL REPRODUCTION.
 !! www.clutchprep.com CONCEPT: BASICS OF MEIOTIC GENETICS Sexual reproduction involves mixing DNA from individuals to produce genetically distinct offspring Beneficial because it allows for genetic diversity
!! www.clutchprep.com CONCEPT: BASICS OF MEIOTIC GENETICS Sexual reproduction involves mixing DNA from individuals to produce genetically distinct offspring Beneficial because it allows for genetic diversity
Lesson Overview Meiosis
 11.4 THINK ABOUT IT As geneticists in the early 1900s applied Mendel s laws, they wondered where genes might be located. They expected genes to be carried on structures inside the cell, but which structures?
11.4 THINK ABOUT IT As geneticists in the early 1900s applied Mendel s laws, they wondered where genes might be located. They expected genes to be carried on structures inside the cell, but which structures?
AP Biology Fall Semester Set 1
 1. During which stage does DNA replication occur? A. Prophase B. Metaphase C. Anaphase D. none of these 2. At what phase in the cell cycle does DNA replication occur? A. G1 B. S C. G2 D. M 3. Which of
1. During which stage does DNA replication occur? A. Prophase B. Metaphase C. Anaphase D. none of these 2. At what phase in the cell cycle does DNA replication occur? A. G1 B. S C. G2 D. M 3. Which of
Unit 2: Cellular Chemistry, Structure, and Physiology Module 5: Cellular Reproduction
 Unit 2: Cellular Chemistry, Structure, and Physiology Module 5: Cellular Reproduction NC Essential Standard: 1.2.2 Analyze how cells grow and reproduce in terms of interphase, mitosis, and cytokinesis
Unit 2: Cellular Chemistry, Structure, and Physiology Module 5: Cellular Reproduction NC Essential Standard: 1.2.2 Analyze how cells grow and reproduce in terms of interphase, mitosis, and cytokinesis
Sexual Reproduction and Meiosis. Outline. Random?? fertilization. Chapter 13
 Sexual Reproduction and Meiosis Chapter 13 Outline Reduction Division Unique Features of Meiosis Prophase I Metaphase I Completing Meiosis Second Meiotic Division Sexual Reproduction Origin and Maintenance
Sexual Reproduction and Meiosis Chapter 13 Outline Reduction Division Unique Features of Meiosis Prophase I Metaphase I Completing Meiosis Second Meiotic Division Sexual Reproduction Origin and Maintenance
MEIOSIS CELL DIVISION Chapter
 Section 6.1: Meiosis MEIOSIS CELL DIVISION Chapter 6.1 6.2 WHAT DETERMINES WHAT YOU LOOK LIKE? Meiosis Animation Meiosis creates 4 genetically different gametes (haploid) Mitosis creates 2 identical daughter
Section 6.1: Meiosis MEIOSIS CELL DIVISION Chapter 6.1 6.2 WHAT DETERMINES WHAT YOU LOOK LIKE? Meiosis Animation Meiosis creates 4 genetically different gametes (haploid) Mitosis creates 2 identical daughter
STUDY UNIT 1 MITOSIS AND MEIOSIS. Klug, Cummings & Spencer Chapter 2. Morphology of eukaryotic metaphase chromosomes. Chromatids
 STUDY UNIT 1 MITOSIS AND MEIOSIS Klug, Cummings & Spencer Chapter 2 Life depends on cell division and reproduction of organisms. Process involves transfer of genetic material. New somatic (body) cells
STUDY UNIT 1 MITOSIS AND MEIOSIS Klug, Cummings & Spencer Chapter 2 Life depends on cell division and reproduction of organisms. Process involves transfer of genetic material. New somatic (body) cells
 Question 1: What is the average cell cycle span for a mammalian cell? The average cell cycle span for a mammalian cell is approximately 24 hours. Question 2: Distinguish cytokinesis from karyokinesis.
Question 1: What is the average cell cycle span for a mammalian cell? The average cell cycle span for a mammalian cell is approximately 24 hours. Question 2: Distinguish cytokinesis from karyokinesis.
The Cell Cycle. Chapter 12
 The Cell Cycle Chapter 12 Why are cells small? As cells get bigger they don t work as well WHY? Difficulties Larger Cells Have: More demands on its DNA Less efficient in moving nutrients/waste across its
The Cell Cycle Chapter 12 Why are cells small? As cells get bigger they don t work as well WHY? Difficulties Larger Cells Have: More demands on its DNA Less efficient in moving nutrients/waste across its
Meiosis and Sexual Life Cycles
 Chapter 13 Meiosis and Sexual Life Cycles PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from
Chapter 13 Meiosis and Sexual Life Cycles PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from
Biology. Chapter 12. Meiosis and Sexual Reproduction. Concepts and Applications 9e Starr Evers Starr. Cengage Learning 2015
 Biology Concepts and Applications 9e Starr Evers Starr Chapter 12 Meiosis and Sexual Reproduction 12.1 Why Sex? In asexual reproduction, a single individual gives rise to offspring that are identical to
Biology Concepts and Applications 9e Starr Evers Starr Chapter 12 Meiosis and Sexual Reproduction 12.1 Why Sex? In asexual reproduction, a single individual gives rise to offspring that are identical to
